While radiation treatment for prostate cancer has become much more precise over the years, the organ’s location immediately next to the rectal wall can expose the rectum to high dose radiation, which can lead to disruption in bowel function for some patients.
A new method for reducing these short- and long-term side effects is SpaceOar hydrogel, now available at Fox Chase.
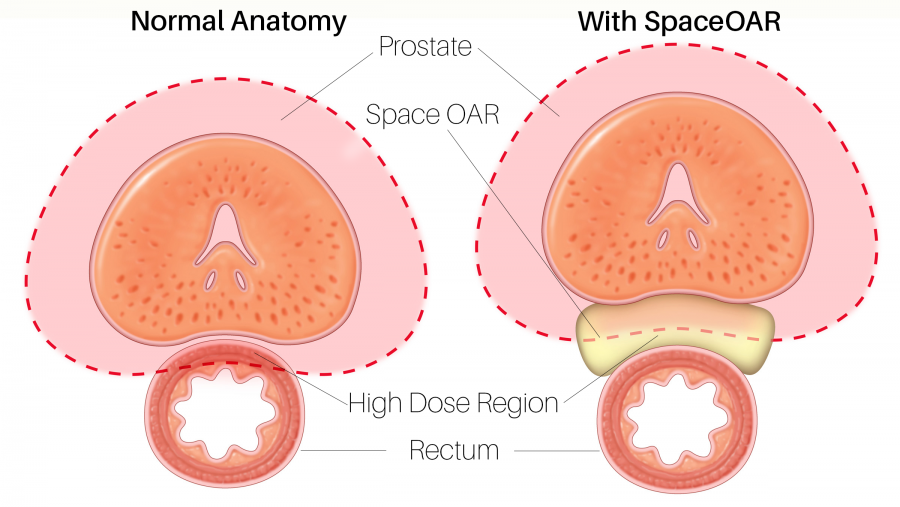
In a minimally invasive outpatient procedure, the two liquid components of SpaceOAR hydrogel are injected through the peritoneum (transperineally) into the space between the rectal wall and the prostate by a urologist. The procedure is done under anesthesia with ultrasound guidance.
The resulting interaction between the two fluids creates a temporary and absorbable gel “spacer” that is primarily made of water. Hydrogels have been used in other implants such as surgical sealants used in the eye, brain and spine.
The “OAR” in SpaceOAR stands for “Organ At Risk,” referring to the rectum.

“The position of the prostate poses particular challenges for the higher doses of radiation treatment required for this type of cancer,” Fox Chase radiation oncologist Mark Hallman MD, PhD said. “The posterior prostate lies right on top of the interior rectal wall and this puts the rectum at increased risk.”
SpaceOAR was approved as a medical device by the FDA in 2015 and Fox Chase is among the first in the Philadelphia area to offer it.
A prospective, randomized, multi-center clinical trial in the United States reported that patients treated with SpaceOAR hydrogel prior to prostate cancer radiation treatment demonstrated bowel, urinary, and sexual benefits through a median of three years of follow-up. The study also found that patients who did not receive SpaceOAR hydrogel experienced a clinically significant decline in bowel, urinary, and sexual quality of life eight times more often than patients who received SpaceOAR hydrogel.
While it is not appropriate for every patient, it can be especially helpful for some men whose abdominal girth, pacemakers, defibrillators, medications, or hip replacements disqualify them from receiving fiducial markers or Calypso beacons to aid in focusing the radiation, Hallman noted.
How it works
“The gel pushes the rectal wall out of the way by an average of 13 millimeters, which is a huge advantage,” Hallman said. "It can deliver radiation to the prostate using our very accurate radiation delivery techniques and avoid affecting the rectal wall.”
The gel remains fully in place for up to three months, allowing for a series of radiation treatments before it is gradually absorbed and cleared through the urine over about six months. Most prostate treatment courses are under one month long. The longest time frame for radiation treatment is about two months, which gives doctors plenty of time to do the treatment planning, Hallman said.
There is a one-week waiting period after the implant to allow the anatomy to settle before proceeding with radiation treatment. SpaceOAR hydrogel can be used with all types of external beam radiation therapy, as well as internal radiation therapy and stereotactic body radiation therapy (SBRT) .
Benefits
The SpaceOar hydrogel has an added benefit of stabilizing the prostate during a treatment session, decreasing prostate motion from bladder filling and rectal gas, Hallman said.
Fox Chase surgical oncologist Richard E. Greenberg, MD, FACS, said the technology is an additional weapon in Fox Chase’s skilled clinicians’ arsenal of approaches for fighting prostate cancer. “Our multi-disciplinary team selects the best treatment for the individual patient based on their needs,” Greenberg said.
What is it made of and is it safe?

SpaceOAR hydrogel is made up of two liquids that, when combined, form a temporary, soft gel material that is mostly made of water - hence the name, hydrogel. It can be safely used in the body without causing injury or a reaction.
Clinical test results
In the US, 222 prostate cancer patients participated in a multi-center, randomized, patient-blinded clinical study. Results showed that the SpaceOAR hydrogel was safe and effective. The average participant gained 1/2 inch of space between the prostate and rectum and had significant reduction in radiation dose to the rectum, resulting in significantly fewer rectal side effects.
In a follow-up study three years after treatment, participants were asked to report on their quality of life for bowel, urinary and sexual functions. These patient-reported outcomes showed that SpaceOAR hydrogel patients experienced significantly fewer long-term rectal side effects, were more likely to maintain sexual function, and had significantly higher patient reported scores for urinary and bowel quality of life.
Risks associated with implantation of SpaceOAR hydrogel
In addition to the risks associated with any medical procedure, potential complications include but are not limited to the following: pain or discomfort associated with SpaceOAR hydrogel; needle penetration of the bladder, prostate, rectal wall, rectum or urethra; injection of SpaceOAR hydrogel into bladder, prostate, rectal wall, rectum or urethra; local inflammatory reactions; infection; injection of air, fluid or SpaceOAR hydrogel intravascularly; urinary retention; rectal mucosal damage, ulcers, necrosis; bleeding, constipation; and rectal urgency.