Related Articles
00 / 00

Request an Appointment
Chief, Division of Sarcoma Medical Oncology
Vice Chair, Department of Hematology/Oncology
Physician Director, Clinical Trials Office
Associate Director, Clinical Research
Professor, Department of Hematology/Oncology
NCCN, Adolescent and Young Adult Oncology Panel Member
NCCN, Soft Tissue Sarcoma Panel Member
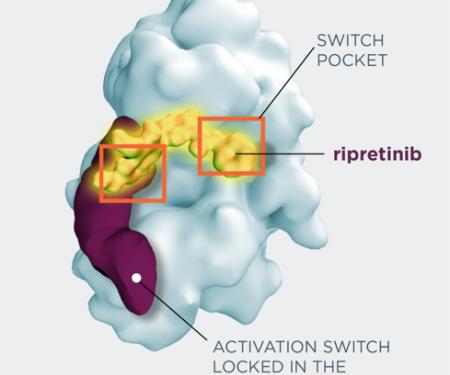
Uterine Sarcoma, GIST (Gastrointestinal Stromal Tumor), Skin Cancer, Sarcoma, Melanoma
Chemotherapy; tyrosine kinase therapy; immunotherapy
I first had the opportunity to treat patients with sarcoma during my oncology fellowship training. I really enjoyed the challenge of this area of oncology because of the many types of tumors and their rarity. When I meet a patient, I work to be certain the pathologic diagnosis is correct and address what we need to do to treat the disease. I collaborate with Fox Chase pathologists, radiologists, surgeons, radiation oncologists and social workers to develop the right treatment plan. We meet as a team weekly at the Sarcoma multi-disciplinary tumor board to review individual patient cases and agree on the best management.
I am also committed to developing new therapies for patients with sarcomas. The strides made in improving cancer care have come from research and clinical trials. I work closely with scientists to better understand gastrointestinal stromal tumors and other sarcomas. I aim to have research studies as a treatment option for my sarcoma patients.
I’m a 61-year-old professional singer-songwriter.

I recently ended a long-term relationship with Fox Chase Cancer Center, where I have been receiving treatment on and off for more than a decade.

When I met the physicians at Fox Chase Cancer Center who would be treating me, I knew right away there were no better hands to be in.
I am a breast cancer survivor, but my second journey with cancer started about two and a half years ago, when I started to experience two symptoms. First, I was waking every two hours during the night to empty my bladder. Second, I started having quite a bit of trouble moving my bowels. I put off seeing a doctor about these symptoms, but finally a friend of mine suggested that I go see a gynecologist to discuss what was happening.

In 2021, I started having pain in my left leg. I visited my family doctor, and at first we thought it might be a deep vein thrombosis. When we ruled that out, I saw a vascular doctor in case it might be varicose veins. That doctor had me get an MRI, and after those results came back, I had to get a biopsy. That’s when the doctors confirmed that I had a stage IV tumor in my leg.
Hensley H, Devarajan K Johnson JR, Piwnica-Worms D, Godwin AK, von Mehren M, Rink L. Evaluating New Therapies in Gastrointestinal Stromal Tumor Using in vivo Molecular Optical Imaging. Cancer Biology and Therapeutics, 15(7):911-8, 2014. PubMed
Demetri G, Reichardt P, Kang Y-K, Blay JY, Rutkowski P, Gelderblom H, Hohenberger P, Leahy M, von Mehren M, Joensuu H, Badalamenti G, Blackstein M, Le Cesne A, Schffski P, Maki RG, Bauer S, Nguyen BB, Xu J, Nishida T, Chung J, Kappeler C, Kuss I, Laurent D, Casali PG. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet, 381(9863):295-302. 2013. PubMed PMID: 23177515; PubMed Central PMCID: PMC3819942.
Belinsky MG, Rink L, Flieder DB, Jahromi MS, Schiffman JD, Godwin AK, von Mehren M. Overexpression of insulin-like growth factor 1 receptor and frequent mutational inactivation of SDHA in wild-type SDHB-negative gastrointestinal stromal tumors. Genes, Chromosomes and Cancer, 52(2):214-24, 2013. PubMed PMID: 23109135; PubMed Central PMCID: PMC3564228.
George S, Wang Q, Heinrich MC, Corless CL, Zhu M, Butrynski JE, Morgan JA, Wagner AJ, Choy E, Tap WD, Yap JT, Van den Abbeele AD, Manola JB, Solomon SM, Fletcher JA, von Mehren M, Demetri GD. Efficacy and safety of regorafenib in patients with metastatic and/or unresectable gastrointestinal stromal tumor (GIST) following failure of imatinib and sunitinib: a multicenter phase II trial. Journal of Clinical Oncology, 30(19):2401-7, 2012.
von Mehren M, Rankin C, Goldblum JR, Demetri GD, Bramwell V, Ryan CW, Borden E. Phase II SWOG-directed Intergroup Trial (S0505) of Sorafenib in Advanced Soft Tissue Sarcomas. Cancer, 118(3):770-6, 2012.
Demetri GD, Wang Y, Wehrle E, Racine, Nikolova Z, Blanke C, Joensuu H, von Mehren M. Imatinib Plasma Levels Correlate With Clinical Benefit in Patients With Unresectable/ Metastatic Gastrointestinal Stromal Tumors (GIST). Journal of Clinical Oncology, 27(19):3141-7, 2009. Epub May 18 2009. PubMed PMID: 18942073; PubMed Central PMCID: PMC2606912.
Eisenberg BL, Harris J, Blanke CD, Demetri GD, Heinrich MC, Watson JC, Hoffman JP, Okuno S, Kane JM, von Mehren M. Phase II trial of Neoadjuvant/Adjuvant Imatinib Mesylate for Advanced Primary and Metastatic/Recurrent Operable Gastrointestinal Stromal Tumors (GIST) – early results of RTOG 0132. Journal of Surgical Oncology, 99(1):42-7, 2009. PubMed
The following ratings and reviews are based on verified feedback collected from independently administered patient experience surveys. The ratings and comments submitted by patients reflect their own views and opinions. Patient identities are withheld to ensure confidentiality and privacy. Learn more about our Patient Experience Ratings.




















Patient comments
Excellenex
Dr Von Mehren is an outstanding Doctor
Dr. Von Mueren is a kind, thoughtful and very thorough provider. She's very professional and I like her very much as my doctor.
Outstanding
Dr Von Mehren is outstanding-she is doing her best to keep me alive-My wife and I love her