Breadcrumb
- Home
- Jennifer Rhodes
Jennifer Rhodes, PhD

Director, Research Regulatory Affairs
Biosafety Officer
Assistant Research Professor
Director, Zebrafish Facility
Lab Overview
Zebrafish (Danio rerio) serve as a powerful vertebrate model for dissecting the molecular and cellular mechanisms underlying cancer and normal cell development. Their optical transparency, rapid embryogenesis, and genetic tractability enable real‑time visualization of oncogenic processes and cell lineage specification across developmental stages. Using transgenic reporter lines, CRISPR-based genome editing, and cell-based xenograft methodologies, our research program investigates how dysregulated signaling networks drive malignancy and perturb normal embryogenesis. These studies enhance New Approach Methodologies (NAMs) using a non-mammalian vertebrate model, advancing innovative strategies that increase biological relevance and efficiency while reducing reliance on traditional mammalian models. Our zebrafish research program aims to accelerate discovery and deepen understanding of pathways central to cancer biology and healthy development. The zebrafish facility supports all research using this model at Fox Chase Cancer Center; contact the Rhodes lab staff for more information.
Educational Background
- PhD, Oregon Health Sciences University, Portland, OR
- BS, Oregon State University, Corvallis, OR
Memberships
- American Biological Society Association
People
Additional Staff
- Bruce Young, Volunteer
Research Facility
Research Interests
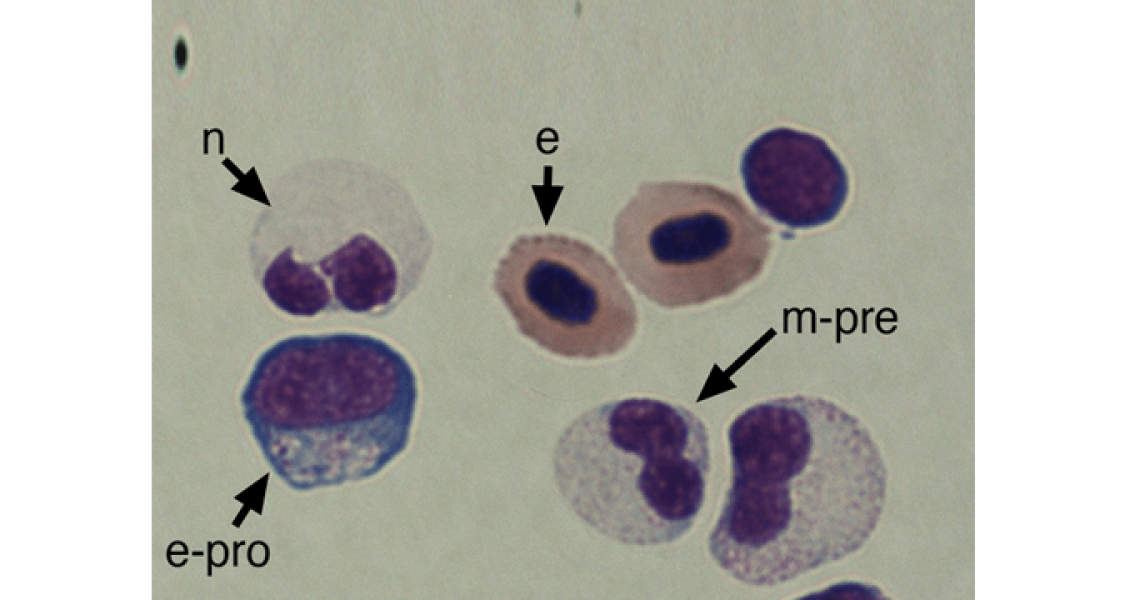
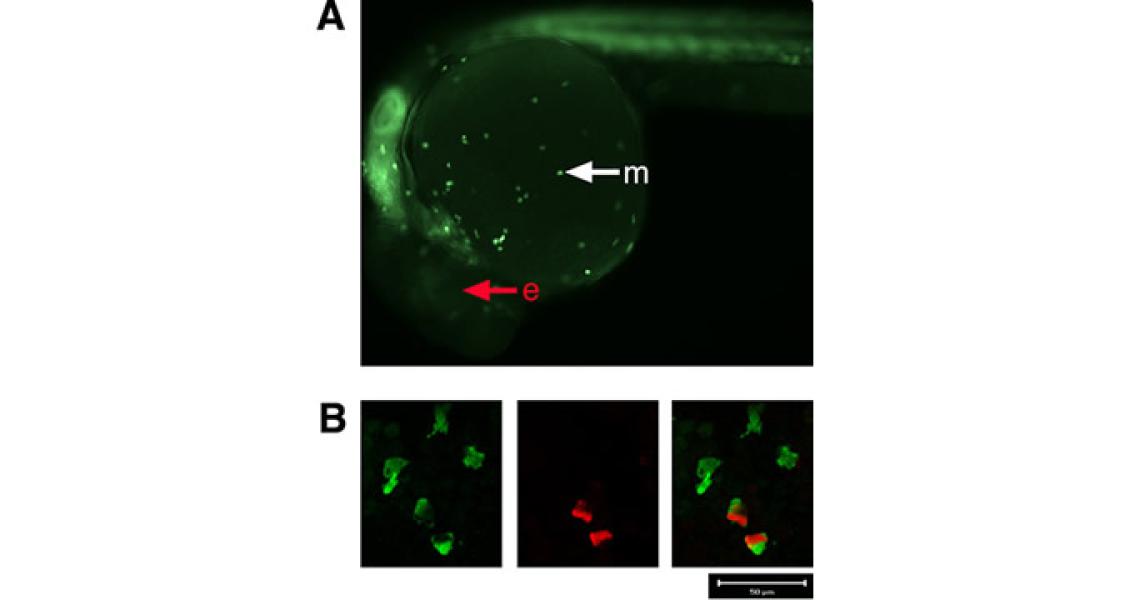
Healthy development including hematopoiesis in zebrafish
Cancer-relevant zebrafish model development
Biosafety in animal and lab science
Selected Publications
Araiza-Olivera D, Feng Y, Semenova G, Prudnikova TY, Rhodes J, Chernoff J. Suppression of RAC1-driven malignant melanoma by group A PAK inhibitors. Oncogene, 37(7):944-52, 2018. PMC5814328
Seiler C, Gebhart N, Zhang Y, Shinton SA, Li Y, Ross NL, Liu X, Li Q, Bilbee AN, Gaurav K. Varshney GK, LaFave MC, Burgess SM2, Balciuniene J, Balciunas D, Hardy RR, Kappes DJ, Wiest DL, Rhodes J. Mutagenesis screen identifies agtpbp1 and eps15L1 as essential for T lymphocyte development in zebrafish. Plos One. 2015 Jul 10;10(7):e0131908. doi: 10.1371/journal.pone.0131908. PubMed PMCID: PMC4498767.
Pimtong W, Datta M, Ulrich AM, Rhodes J. Drl.3 governs primitive hematopoiesis in zebrafish. Sci Rep. 2014 Jul 23;4:5791. PubMed PMCID: PMC4107348.
Zhang Y, Duc AC, Rao S, Sun XL, Bilbee AN, Rhodes M, Li Q, Kappes DJ, Rhodes J, Wiest DL. Control of hematopoietic stem cell emergence by antagonistic functions of ribosomal protein paralogs. Dev Cell. 2013 Feb 25;24(4):411-25. PubMed PMCID: PMC3586312.
Robu ME, Zhang Y, Rhodes J. Rereplication in emi1-deficient zebrafish embryos occurs through a Cdh1-mediated pathway. PLoS One. 2012;7(10):e47658. PubMed PMCID: PMC3474755.
Payne EM, Bolli N, Rhodes J, Abdel-Wahab OI, Levine R, Hedvat CV, Stone R, Khanna-Gupta A, Sun H, Kanki JP, Gazda HT, Beggs AH, Cotter FE, Look AT. Ddx18 is essential for cell-cycle progression in zebrafish hematopoietic cells and is mutated in human AML. Blood. 2011 Jul 28;118(4):903-15. PubMed PMCID: PMC3148170.
Bolli N, Payne EM, Rhodes J, Gjini E, Johnston AB, Guo F, Lee JS, Stewart RA, Kanki JP, Chen AT, Zhou Y, Zon LI, Look AT. cpsf1 is required for definitive HSC survival in zebrafish. Blood. 2011 Apr 14;117(15):3996-4007. PubMed PMCID: PMC3087528
Robinson BW, Germano G, Song Y, Abrams J, Scott M, Guariento I, Tiso N, Argenton F, Basso G, Rhodes J, Kanki JP, Look AT, Balice-Gordon RJ, Felix CA. mll ortholog containing functional domains of human MLL is expressed throughout the zebrafish lifespan and in haematopoietic tissues. Br J Haematol. 2011 Feb;152(3):307-21. PubMed PMCID: PMC3158705.
Rhodes J, Amsterdam A, Sanda T, Moreau LA, McKenna K, Heinrichs S, Ganem NJ, Ho KW, Neuberg DS, Johnston A, Ahn Y, Kutok JL, Hromas R, Wray J, Lee C, Murphy C, Radtke I, Downing JR, Fleming MD, MacConaill LE, Amatruda JF, Gutierrez A, Galinsky I, Stone RM, Ross EA, Pellman DS, Kanki JP, Look AT. Emi1 maintains genomic integrity during zebrafish embryogenesis and cooperates with p53 in tumor suppression. Mol Cell Biol. 2009 Nov;29(21):5911-22. PubMed PMCID: PMC2772726.
Rhodes J, Hagen A, Hsu K, Deng M, Liu TX, Look AT, Kanki JP. Interplay of pu.1 and gata1 determines myelo-erythroid progenitor cell fate in zebrafish. Dev Cell. 2005 Jan;8(1):97-108. PubMed.
Hsu K, Traver D, Kutok JL, Hagen A, Liu TX, Paw BH, Rhodes J, Berman JN, Zon LI, Kanki JP, Look AT. The pu.1 promoter drives myeloid gene expression in zebrafish. Blood. 2004 Sep 1;104(5):1291-7. PubMed.